As more vehicles transition to electric power, the need for lithium-ion batteries continues to increase.
However, the cost of these batteries is an issue, according to University of Texas at Dallas professor of materials science and engineering Dr. Kyeongjae Cho.
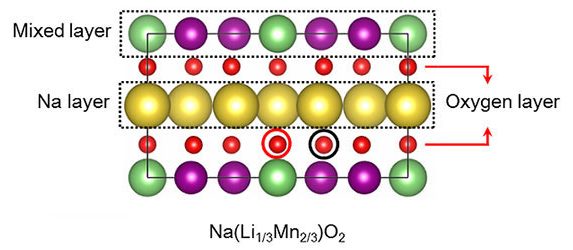
The research team’s sodium-ion design, which retains the high energy density of a lithium-ion cathode, replaces most of the lithium atoms (green) with sodium (yellow). The layered structure of the new material also incorporates manganese (purple) and oxygen (red). [Photo: UTD]
“Lithium is a more expensive, limited resource that must be mined from just a few areas on the globe,” Cho said in a release.
Cho and his team, in collaboration with Seoul National University, have developed a new material that could offer a lower-cost and an eco-friendlier option to replace current battery technology.
Researchers first sought to substitute lower-costing sodium for most of the lithium component in batteries.
Initially, using sodium had its drawbacks, providing a 20 percent lower energy density compared to lithium batteries. The energy density regulates a battery’s life.
By combining sodium with manganese, the UTD researchers able to make up the remaining energy difference.
“Our sodium-ion material is more stable, but it still maintains the high energy capacity of lithium.”
Dr. Kyeongjae Cho
In a standard lithium battery, a positive electrode and a negative electrode are present on opposite sides with an electrolyte in-between. The positive electrode consists of lithium, cobalt, nickel, and oxygen, while the negative is comprised of graphite. The UTD design replaces majority of the lithium atoms with sodium and implements manganese instead of cobalt and nickel.
Cho and his research team took this knowledge and have used computer simulations to research the most successful arrangement of atoms before they begin making and testing materials.

Dr. Kyeongjae Cho [Photo: UTD]
“Our sodium-ion material is more stable, but it still maintains the high energy capacity of lithium,” Cho said. “And we believe this is scalable, which is the whole point of our research. We want to make the material in such a way that the process is compatible with commercial mass production.”
This isn’t Cho’s first foray into discovering alternative makeups for batteries.
Last May, Cho published research about new “catalyst materials” for lithium-air batteries that he said could last five times longer than current lithium-air batteries on the market.
Dallas Innovates, every day

























