clinical trials
Alliance Clinical Network Names Former Ascend Aesthetic CEO as Its New Chief Executive


Marc Chamberlain, M.D., has been appointed as the Chief Medical Officer at Starlight Therapeutics.
Starlight Therapeutics is a Dallas-based Lantern Pharma subsidiary focused exclusively on the clinical development of therapies for central nervous system and brain cancers with limited or no effective therapeutic options.
In his role as CMO, Chamberlain will oversee the company’s upcoming clinical trials focused on several serious and complex brain-related conditions: glioblastoma and other high-grade gliomas (aggressive types of brain tumor), brain metastases in adults (secondary brain tumors originating from cancer elsewhere in the body), and two types of pediatric brain tumors — atypical teratoid rhabdoid tumors (ATRT) and diffuse intrinsic pontine glioma (DIPG)....
The Last Word: Nicole Small On Why the Dallas-Based ARPA-H Hub Is Focused on Clinical Trials





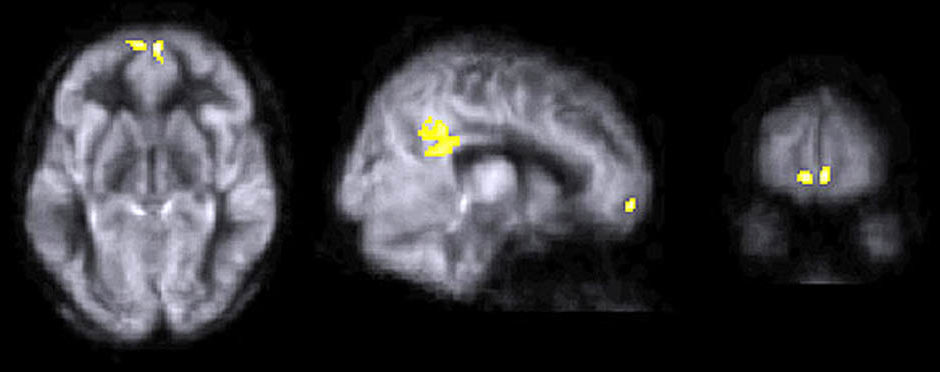
This image highlights brain regions that showed significant cerebral blood flow increases in the cognitive training group. [Photo courtesy of Center for BrainHealth]
![Anthony Milonas, CEO of Alliance Clinical Network. [Photo: LinkedIn]](https://s24806.pcdn.co/wp-content/uploads/2025/11/AllianceClinicalNetwork_AnthonyMilonas-970-1-940x436.jpg)




















