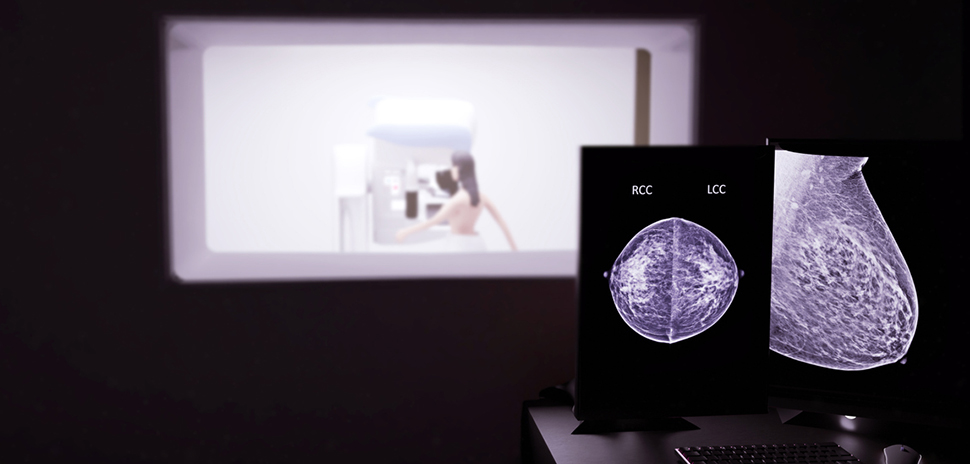
Perimeter Medical Imaging AI—a commercial-stage medical technology company with a U.S. office in Dallas and headquarters in Toronto—has received U.S. Food and Drug Administration premarket approval for Claire, the first AI-enabled imaging device approved in the United States for intraoperative breast cancer margin assessment.
Designated by the FDA as a Breakthrough Device, Claire is designed to enhance surgeons’ ability to detect difficult-to-see cancer during breast-conserving surgery and potentially reduce the need for re-operations.
“Repeat breast cancer surgeries due to residual disease remain a significant clinical, health, and economic burden,” Perimeter CEO Adrian Mendes said in a statement. “Claire’s FDA approval marks a major milestone in breast cancer care, as we advance our goal of reducing repeat surgeries so that no patient has to be told ‘we didn’t get it all.'”
“We plan to begin a nationwide launch in the coming weeks,” Mendes added, “so that surgeons can rapidly adopt the industry’s first FDA-approved real-time AI-powered imaging technology for breast cancer surgery.”
Improving the level of care
Claire combines Perimeter’s proprietary AI with its patented wide-field OCT imaging to allow for high-resolution, real-time evaluation of excised tumor margins, the company said. The system provides 10x higher resolution than standard X-ray and ultrasound at 2mm imaging depth—the clinically relevant margin width for breast cancer margin assessment. Claire’s innovative AI technology was trained on Perimeter’s proprietary and growing OCT image library of over 2 million breast tissue images.
Today, surgeons use physical examination, and in limited cases, intraoperative pathology to assess margins before sending specimens to pathology for final evaluation. The process means that patients may wait up to a week or more to learn whether margins are clear or additional surgery is required, Perimeter noted.
Claire is designed to identify areas of concern during surgery, helping surgeons determine whether to remove more tissue before completing the procedure. The current national average indicates that repeat surgeries occur in about 20% of breast-conserving surgeries in the U.S, Perimeter said.
“Despite progress in breast cancer treatment, intra-operative margin assessment remains challenging, often leading to excess removal of healthy tissue, re-operations, and anxiety for patients as they await pathology results,” said Dr. Alastair Thompson, Surgeon and Professor, Section Chief of Breast Surgery, Olga Keith Wiess Chair of Surgery at Baylor College of Medicine, Breast Cancer Program Leader at the Dan L Duncan Comprehensive Cancer Center, and the primary principal investigator of the pivotal trial that supported Claire’s PMA application.
“Claire has the potential to become a new standard in breast surgical care helping reduce re-excisions while improving patient outcomes,” Thompson added.
Claire’s trial and potential
During the trial period, Claire demonstrated an 88.1% margin accuracy and a statistically significant reduction in patients with residual cancer post-surgery compared to the standard of care alone.
The large-scale clinical trial was funded in part by a grant of $7.4 million from the Cancer Prevention and Research Institute of Texas (CPRIT).
A key component of Claire is its AI engine, trained on millions of proprietary OCT images of cancerous and healthy tissue. The image library is generated using Perimeter’s patented OCT imaging engine. Every surgical procedure performed with Claire generates new data that can be used to improve the AI product, helping to create better outcomes for future patients, the company said.
Claire is one of the few AI-enabled class III devices on the U.S. market today, Perimeter said. The PMA approval includes planned AI enhancements that can be implemented without additional FDA interaction.
“Developing AI for clinical use is incredibly challenging, requiring large, high-quality datasets that reflect real-life patient diversity,” said Perimeter’s Chief Innovation Officer and co-founder Andrew Berkeley. “This approval reflects years of hard work with our clinical partners integrating AI directly into surgical decision-making and advancing its role from operational support to real-time patient care.”
‘The starting line of something truly transformational’
Claire’s FDA approval represents Perimeter’s first commercialization of its proprietary OCT-AI platform. The company said that its patented wide-field OCT technology, proprietary dataset, and integrated AI capabilities target an estimated 300,000 annual U.S. breast cancer surgeries, while providing a scalable foundation for expansion into additional cancer surgeries, biopsy procedures, and pathology applications.
Chamath Palihapitiya, Silicon Valley venture capitalist, entrepreneur, and former Facebook executive, said, “I’ve been a major investor in Perimeter for several years, and it’s been the incredible clinical and commercial potential of Claire that has driven my excitement all along. After rigorous development, clinical and regulatory engagement, and collaboration with the medical community, I believe Perimeter is at the starting line of something truly transformational.”
“Claire delivers greater peace of mind,” Palihapitiya added in a statement. “Both for the surgeon who currently faces nearly 1-in-5 odds of needing to perform repeat surgery due to positive margins, and for the breast cancer patient, who, under the current paradigm, typically has to wait (and worry) for up to 10 days to learn whether an additional surgery is required. A second surgery is an emotional and physical journey that we are trying to prevent. This is exactly the kind of AI-driven innovation that can improve the standard of care by delivering measurable, real-world medical impacts. I’m excited to see what Perimeter can accomplish, as it commercializes the first AI-enabled imaging device for breast cancer surgery.”
Don’t miss what’s next. Subscribe to Dallas Innovates.
Track Dallas-Fort Worth’s business and innovation landscape with our curated news in your inbox Tuesday-Thursday.